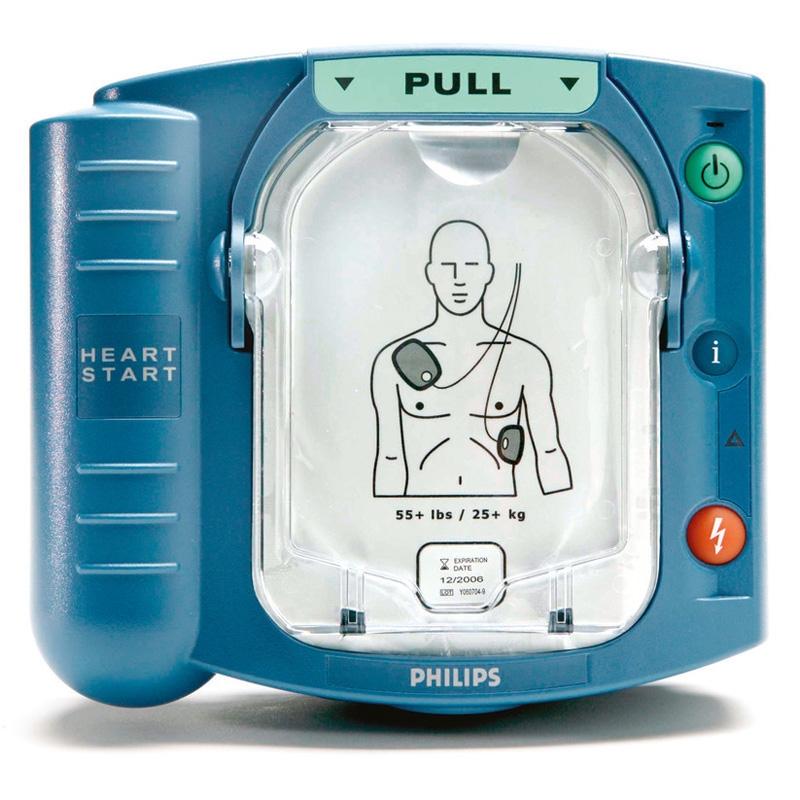
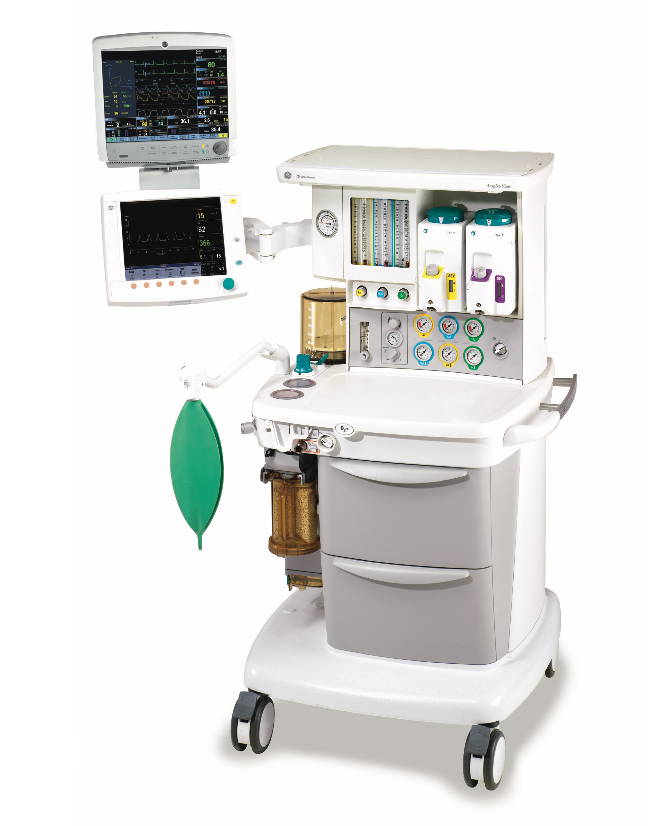
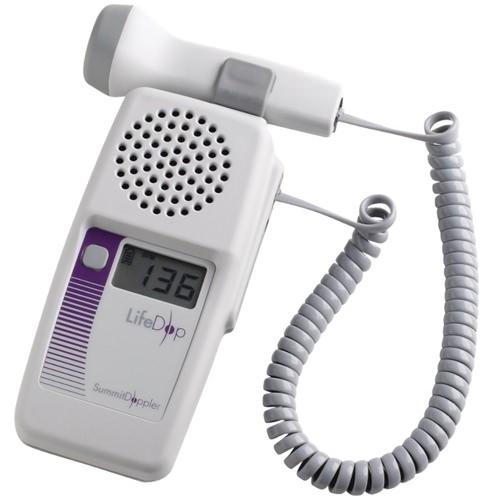
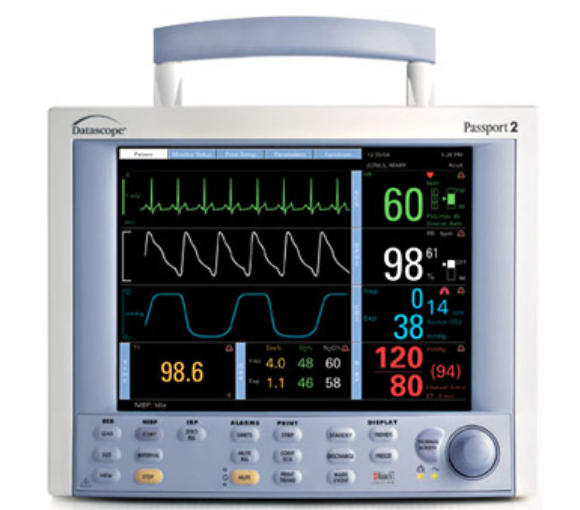
At Integris Equipment, we have partnered with the best brands and manufacturers in the industry in order to supply you with the highest quality products for your medical equipment needs.
Our key partners include well known brand manufacturers such as Zoll, Welch Allyn, Masimo, Medtronic (Covidien) and Summit Doppler. For the full range of medical equipment manufacturers we work with, and to view their product ranges, please click here.